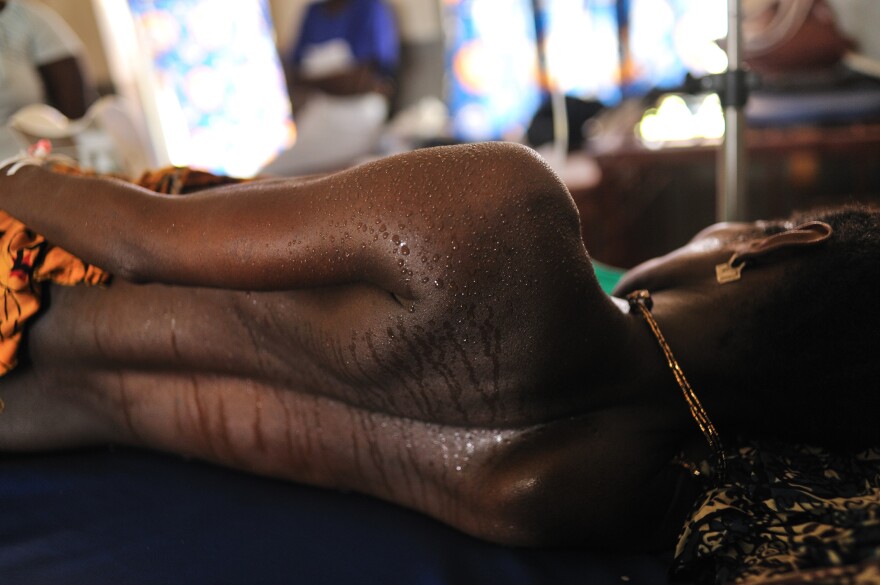
One of the indirect effects of the Ebola epidemic that tore through West Africa between 2014 and 2016 was the dramatic decline in access to care for pregnancy and childbirth, increasing the risk of injury or death among expectant mothers across the affected zone.
Now experts worry the novel coronavirus could have the same effect in poor countries around the world, worsening a global maternal mortality rate the World Health Organization has described as "unacceptably high."
In a study published in May in the Lancet Global Health, researchers at the Johns Hopkins Bloomberg School of Public Health said the spillover effects of COVID-19 could result in an additional 56,700 maternal deaths over the next six months in 118 low- and middle-income countries.
Even in the best of times, these countries account for an estimated 94% of the 295,000 maternal deaths recorded annually worldwide, the bulk of them in sub-Saharan Africa. The leading cause of those deaths is what's known as postpartum hemorrhage, or excessive blood loss after the birth of a baby. And that's driven in large part by the fact that, for a variety of reasons, millions of women still deliver at home (or en route to a health facility), many of them with no one else present.
Despite early predictions that the coronavirus would wreak havoc in African countries, many have so far managed to limit its spread, offering lessons for the world. Still, the United Nations has raised the alarm about the grave threat to women and girls, predicting that reductions in routine health services and access to contraceptives could result in 7 million unintended pregnancies.
"Left unchecked, these reductions ... will be more catastrophic for mothers and children than COVID-19 itself," wrote Henrietta Fore, executive director of the U.N. Children's Fund, in a mid-May commentary to the Lancet Global Health.
"There may likely be a decrease in facility births," says Jeffrey Smith, deputy director of maternal and child health at the Bill and Melinda Gates Foundation, which is a funder of NPR and of this blog. "That's what we saw at the height of the Ebola epidemic in Guinea, Liberia and Sierra Leone," all places where a woman's risk of bleeding out in childbirth was already very high.
For Smith, the prospect of a similar situation in the months ahead underscores the importance of a controversial intervention he and colleagues have championed for decades: equipping pregnant women in remote rural areas with a drug they can take to prevent postpartum hemorrhage should they deliver at home.
That drug is misoprostol.
An inexpensive, widely available medication, misoprostol reduces blood loss by causing the uterus to contract, compressing the blood vessels that feed the placenta.
But the drug is also well known for its ability to safely end an early pregnancy, and the controversy has stemmed in large part from concerns about the latter — that impoverished women with little control over their reproductive lives might be tempted to use it for abortion.
Though unsupported by the evidence, these fears have long hampered efforts in some countries to make the drug more widely available, say researchers like Ndola Prata, director of the Bixby Center for Population, Health, and Sustainability at the University of California, Berkeley.
"A number of governments have started down this path," says Prata, whose work helped bring about regulatory approval of misoprostol for the prevention of postpartum hemorrhage in countries across sub-Saharan Africa and southeast Asia. "But the extent to which women who need misoprostol have access to it — we're still very far away. We're missing a huge opportunity to save lives."
The history of misoprostol: from ulcers to abortion
Misoprostol's story begins in the mid-1980s, when it came on the market under the brand name Cytotec for the prevention of gastric ulcers.
Soon after its introduction, women in Brazil discovered that the cheap, over-the-counter drug could be safely and discreetly used to induce an abortion. As its popularity grew, physicians there noticed a reduction in the number of women presenting with severe post-abortion complications, and by the mid-1990s, word of misoprostol's abortifacient qualities had spread across Latin America and beyond.
Meanwhile, scientists were learning of misoprostol's many other obstetric applications, including the ability to treat a miscarriage by expelling retained tissue and to prevent postpartum hemorrhage.
When it comes to the latter, misoprostol is not the gold standard; WHO instead recommends oxytocin, which studies have shown to be marginally more effective and to have fewer side effects than misoprostol, which can sometimes cause shivering and fever.
But oxytocin suffers from several crucial limitations: the drug can only be administered via injection, must be given by a skilled provider and requires constant refrigeration. So it isn't feasible for use in places where electricity is unreliable, skilled care is scarce and basic supplies are often out of stock.
Misoprostol, by comparison, comes in a tablet form that can be stored for years at room temperature and taken with a cup of water. Those qualities led the United States Agency for International Development to support programs in poor countries promoting its use at the community level. In 2011 WHO added misoprostol to its Model List of Essential Medicines, recommending it for prevention of postpartum hemorrhage where oxytocin is not available.
The recommendation was a breakthrough for reproductive health groups, who for years had petitioned the health authority to grant the drug its stamp of approval.
Yet in doing so, WHO stopped short of endorsing misoprostol for "self-care," citing a dearth of large-scale evaluations of its advance distribution to women at risk of giving birth at home. And Prata and others say that's had a chilling effect on efforts around the world, leading all but a small handful of countries to curtail their programs or discontinue them altogether.
"The global community continues to urge the WHO to look at this situation and to consider the movement toward self-care," says Smith, who as chief maternal health advisor to USAID's Maternal and Child Survival Program oversaw pilot studies of advance distribution of misoprostol in more than a dozen countries. "There is one person who will 100% of the time be there for the birth, and that's the woman herself. She should be the one holding the drug."
For Prata, WHO's reluctance to endorse advance distribution smacks of paternalism and an inability to appreciate the reality on the ground — a reality she knows all too well.
A native of Angola, she remembers with painful clarity the first patient she lost to postpartum hemorrhage. It was in the capital, Luanda, in 1987, at the height of the country's 27-year civil war, and Prata, then a newly minted physician at the city's main maternity hospital, had just delivered a healthy baby boy.
"Everything was fine," she recalls. "Everyone was happy. And then the disaster started."
Part of the placenta had broken off in the mother's uterus, causing her to hemorrhage. Though Prata and team were able to remove the retained tissue, they couldn't stop the bleeding. "And this was in a hospital," she says. "So you can imagine the risks for women who deliver at home."
For those women, Prata says, "making misoprostol available is the least we can do."
A call for 'urgent actions'
Opponents counter that the distribution of misoprostol perpetuates a dangerous double standard whereby women in developed countries receive high quality care while women in poor countries are left to make do with an inferior alternative.
"Pregnant women in rural Africa are expected to give birth ... with the false assurance they will deliver safely with misoprostol," wrote the authors of a 2011 letter to WHO on behalf of the faith-based health organization Maternal Life International. "We believe there are significant dangers in the unregulated use of misoprostol," they added, claiming that these dangers have been "underplayed by misoprostol advocates whose real agenda is ... unregulated medical abortion."
In a 2018 application to remove misoprostol from WHO's essential medicines list, Petra Sevcikova and Allyson Pollock, public health researchers at Newcastle University in the United Kingdom, outlined what they describe as "crucial gaps in clinical evidence," arguing that efforts to reduce maternal mortality should focus instead on strengthening health systems. Where skilled birth attendants are present and oxytocin is available, they wrote, "oxytocin is more effective in reducing postpartum bleeding."
But increasingly, studies indicate that it often is not more effective.
As far back as 1987, WHO worried that high temperatures during transport and storage in tropical climates might compromise certain essential medicines. A 1993 analysis confirmed those fears, finding a "widespread problem with the stability of ergometrine," then the drug of choice to prevent postpartum hemorrhage.
The report's authors noted that with oxytocin injection there seemed to be "less of a problem," but that the data were "too few to draw definitive conclusions."
Now that problem looms large.
According to a 2016 systematic review of oxytocin quality in low- and middle-income countries, 57% of oxytocin vials tested in Africa failed international specifications. A study in three districts in Ghana found that the quality of oxytocin "is likely a serious problem," and a similar study in five provinces of the Democratic Republic of Congo found evidence of "widespread product degradation."
In March of last year, representatives of WHO, the U.N. Children's Fund and the U.N. Population Fund issued a joint statement outlining "urgent actions" necessary to ensure the availability of quality-assured oxytocin at all assisted births.
"Like vaccines, when oxytocin is not of sufficient quality or when it is not managed in cold chain, the medicine will rapidly degrade and become ineffective," they warned, noting that countries with tropical climates are at particular risk for oxytocin failures.
"Oxytocin is the best for control of postpartum hemorrhage," said Momade Bay Usta, president of the Association of Mozambican Obstetricians and Gynecologists, which partnered with the government on a 2009 pilot project in four of the country's districts. "But unfortunately, Mozambique is a warm country with not enough electricity. So we knew we needed something else."
Usta recalls that when the pilot was getting underway, some in Mozambique's reproductive health community worried that women with little education might take misoprostol before the baby comes out, which can cause the uterus to rupture. "But now we know that's not true — that if we give women good information, they will use it correctly."
And that's backed up by the data: in a 2013 review of misoprostol distribution programs, Smith and colleagues reported that out of more than 12,000 women who had self-administered misoprostol to prevent postpartum hemorrhage, just 7 did so incorrectly and none had an adverse outcome.
Mozambique's ministry of health has since expanded the pilot to the rest of the country, making the distribution of misoprostol the centerpiece of a national strategy to prevent postpartum hemorrhage at home births — the largest and still one of the only such programs in Africa.
"The criticism that community-based distribution of misoprostol for postpartum hemorrhage prevention will increase its use for abortion — that is probably true," says Angel Foster, a professor of global health at the University of Ottawa and one of the world's foremost experts on reproductive health in post-conflict settings. "And I would argue there's a real ethical problem with introducing misoprostol for prevention and treatment of postpartum hemorrhage and not talking about the fact that it can be used for abortion care."
Foster adds that while researchers probably won't be able to conduct the kind of rigorous randomized controlled trials that would allow WHO to recommend misoprostol's advance distribution, "the evidence we have really aligns with what we're seeing on the ground, which is that this is a drug that has multiple purposes and can save women's lives."
With fears that many African countries could face a prolonged outbreak over several years, Prata says the need for misoprostol has never been more urgent, adding that governments should distribute the drug and contraceptives to all households with a pregnant woman. "We have these powerful tools," she says, "but we don't trust women to take care of themselves. And now in many places, they're the only ones who can."
Patrick Adamsis a freelance journalist based in Atlanta. Find him on Twitter@jpatadams
Copyright 2021 NPR. To see more, visit https://www.npr.org. 9(MDAxODc1ODA5MDEyMjg1MDYxNTFiZTgwZg004))